  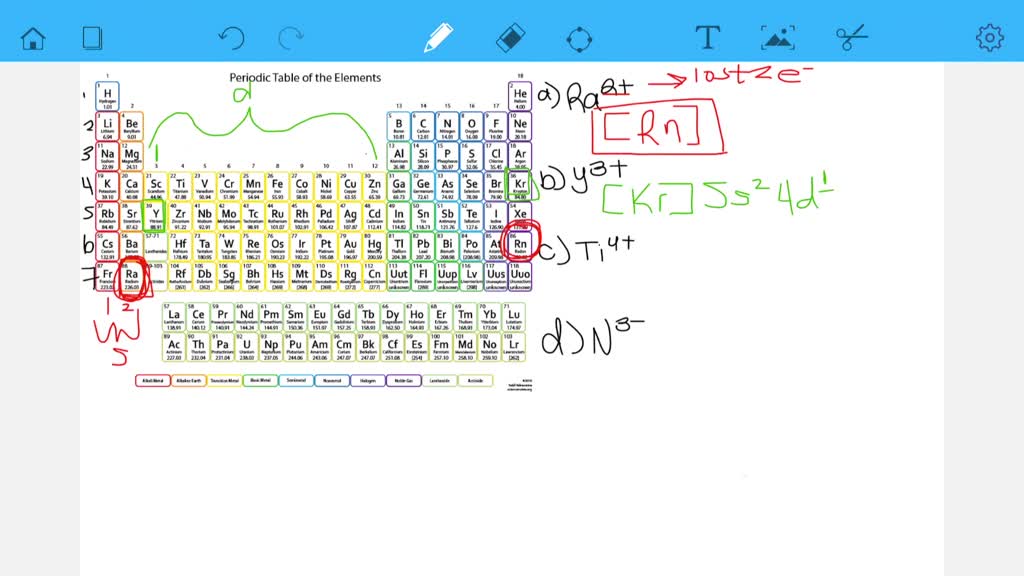
Such an arrangement helps explain the periodicity and periodic trends observed across the elements of the periodic table. 
The N shell containing 4s, 4d, 4p and 4f, can carry 32 electrons. of a state, Energy level above ground state with energy Eand lifetime Dt. The M shell contains 3s, 3p, and 3d, and can carry 18 electrons. Electromagnetic energy will be absorbed or emitted if an electron moves from. The K shell contains a 1s subshell hence it can carry 2 electrons, the L shell has 2s and 2p, and can carry 8 electrons. This decides the electron capacity of the shells. The maximum electrons that can be carried by the sub-shell S is 2, by P is 6, by D is 10, and the F sub-shell can carry 14. Each shell and subshell have a limitation on the amount of electrons that it can carry. The subshells have a distinct shape and configuration, in which the electrons move freely. They stand for sharp (S), principal (P), diffuse (D), and fundamental (F). The shells are labeled K, L, M, N, and so on, from the innermost to the outermost shell.Įach shell has subshells that are named for the type of emission lines produced from different states of angular momentum. With respect to assigning electrons to degenerate p orbitals, Hunds rule. This model has been widely accepted, and according to it, each atom has shells, which further have subshells. For example, the ground state electron configuration of boron (Z 5) is 1s22s22p1. It involves the specific arrangement of electrons in shells and sub-shells of Bohr’s atomic model. The concept of electronic configuration has replaced the older concept of valency and valence electrons. The electronic configuration of each element is decided by the Aufbau principle which states that the electrons fill orbitals in order of increasing energy levels. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
